Health
Study: Scientists Discover Huntington’s Cancer Weapon
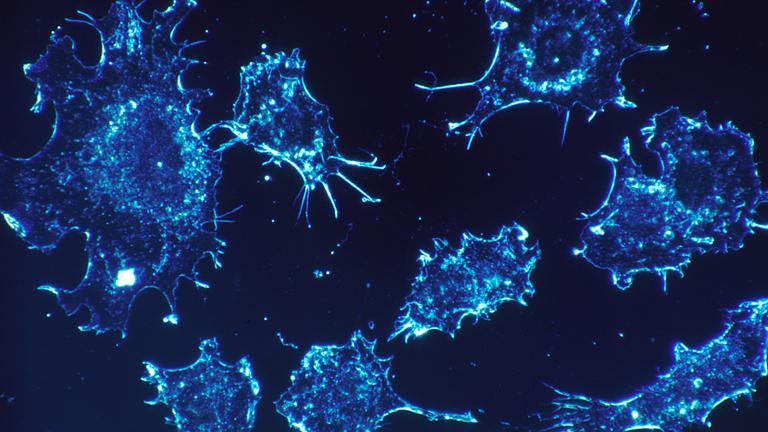
Medical research shows that people diagnosed with Huntington’s disease, an inherited illness that is fatal, are less likely than the general population to develop cancer.
Scientists now have a better understanding of why that is, thanks to the discovery of an “assassin molecule” by Northwestern University scientists.
“This molecule is a super assassin against all tumor cells,” said Marcus Peter, the senior author of a study about the findings, in a press release. “We’ve never seen anything this powerful.”
Huntington’s disease causes the progressive breakdown of nerve cells in the brain, causing death within 10-30 years, according to the Mayo Clinic. It is caused by an overabundance of a specific type of repeating RNA sequences in one gene, huntingtin, present in every cell. Researchers found the defect that causes the disease is also extremely toxic to tumor cells. The repeating small interfering RNA sequences attack genes in the cell that are crucial for survival.
“When you take a gene out that is essential for a cell’s survival, the cell will die,” said Peter, the Tom D. Spies Professor of Cancer Metabolism at Northwestern University Feinberg School of Medicine. Nerve cells in the brain are also vulnerable to this form of death, but cancer cells appear to be much more susceptible. “This is the most toxic (small interfering) RNA that we’ve ever seen. And that at least now, as a concept, provides an explanation as to why these (Huntington’s) patients have reduced (rates of) cancer.”
To test this in a treatment situation, researchers delivered the small interfering RNAs to mice with human ovarian cancer. “The really interesting part was the tumors started to regress and there was no toxicity in the mice, and the tumors did not become resistant (to the treatment),” said Peter.
Andrea Murmann, a research assistant professor in medicine at Feinberg and first author of the study, discovered the Huntington’s cancer weapon and used the molecule to treat human and mouse ovarian, breast, prostate, liver, brain, lung, skin and colon cancer cell lines. The molecule killed cancer cells in both species.
“The question isn’t, ‘Can we kill the cells?’ That’s fundamentally clear. Killing cancer cells is easy,” said Peter. “Is the patient going to survive? That’s the big question.”
More research is needed to understand why cancer cells are targeted and normal cells are protected, Peter said. In addition, scientists need to develop a viable way to deliver the molecules.
“You need an agent that can be stored on the shelf so the physician eventually can take a syringe and inject the agent or give a pill,” he said. Currently, the molecules need to be made “fresh” for each treatment – a process which can take two days.
The discovery stemmed from previous research which identified specific molecules hidden in every cell that trigger a mechanism that causes cancer to commit suicide. “I thought maybe there is a situation where this kill switch is overactive in certain people, and where it could cause loss of tissues,” Murmann said in a press release. “These patients would not only have a disease with an RNA component, but they also had to have less cancer.”
Murmann identified diseases that had a lower rate of cancer and had a suspected contribution of RNA to disease pathology. Huntington’s disease was the most prominent. Analyzing the repeating sequences in huntingtin, the gene that causes the disease, Murmann noticed a similar composition to the kill switch (or suicide molecules) identified by Peter. Both were rich in the C and G nucleotides, two of the molecules that form the building blocks of DNA and RNA (the others are A and T).
“Toxicity goes together with C and G richness,” Murmann said. “Those similarities triggered our curiosity.”
In a person with Huntington’s disease, the gene huntingtin has an overabundance of triplet sequence CAG, said Peter. The longer the repeating sequence, the earlier a person will develop the disease and the more severe it will be.
“We believe a short-term treatment cancer therapy for a few weeks might be possible, where we could treat a patient to kill the cancer cells without causing the neurological issues that Huntington’s patients suffer from,” Peter said. Huntington’s patients have a lifetime exposure to these toxic RNA sequences, but generally don’t develop symptoms of the disease until age 40.
The study was published Monday in the journal of EMBO Reports.
Contact Kristen Thometz: @kristenthometz | [email protected] | (773) 509-5452
Related stories:
 UChicago’s Chuan He Awarded 2017 Paul Marks Prize in Cancer Research
UChicago’s Chuan He Awarded 2017 Paul Marks Prize in Cancer Research
Nov. 10, 2017: The biennial award is bestowed upon three promising researchers under the age of 45 making significant contributions to understanding the disease or improving treatments.
 Scientists Target Cancer’s ‘Achilles’ Heel’ to Stop Therapy Resistance
Scientists Target Cancer’s ‘Achilles’ Heel’ to Stop Therapy Resistance
Nov. 9, 2017: Northwestern University scientists believe they may have found the “Achilles’ heel” of cancer. In a recent study, researchers were able to almost completely eradicate the disease in laboratory cell cultures.
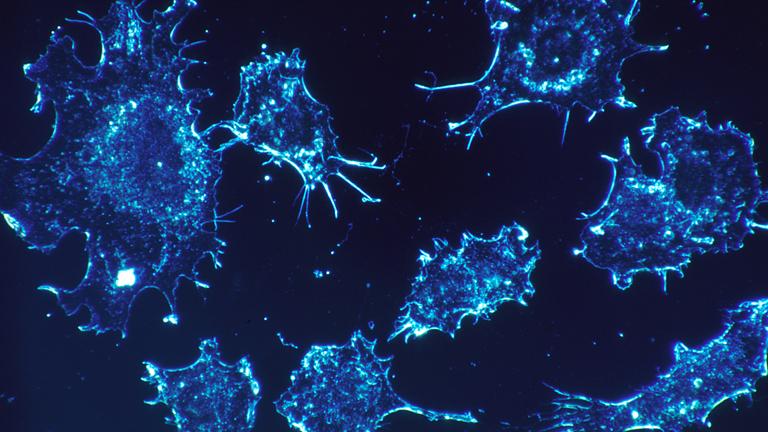 Northwestern Scientists Discover Molecules Capable of Killing Cancer
Northwestern Scientists Discover Molecules Capable of Killing Cancer
Oct. 19, 2017: “Our research may be tapping into one of nature’s original kill switches, and we hope the impact will affect many cancers,” said Northwestern scientist Marcus Peter. “Our findings could be disruptive.”







