Health
FDA, FTC Issue Warnings to Companies Claiming to Treat, Prevent Coronavirus
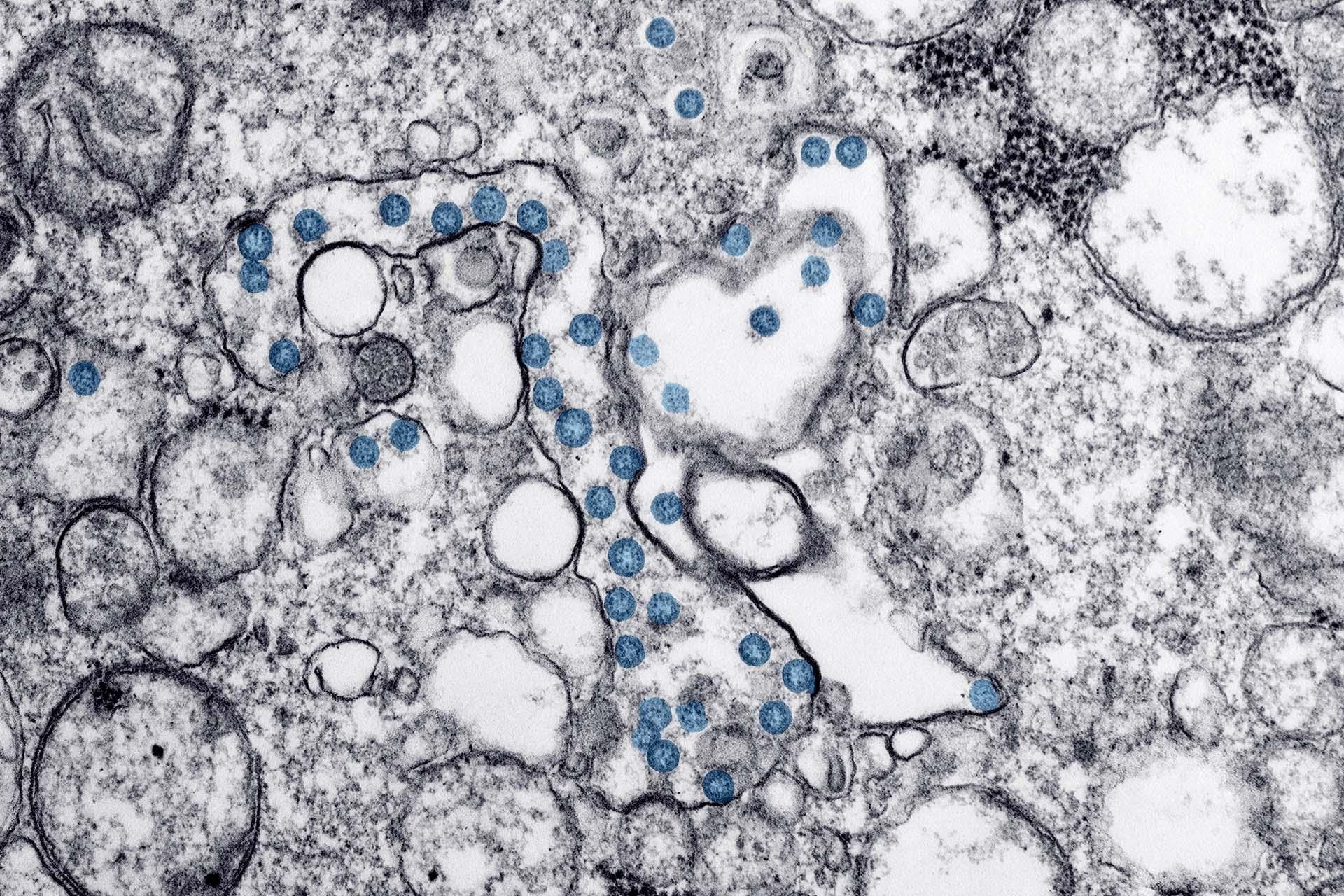 This image from a transmission electron microscopic image shows a sample from the first U.S. case of COVID-19. The spherical viral particles, colored blue, contain cross-sections through the viral genome, seen as black dots. (Image provided by the U.S. Centers for Disease Control and Prevention)
This image from a transmission electron microscopic image shows a sample from the first U.S. case of COVID-19. The spherical viral particles, colored blue, contain cross-sections through the viral genome, seen as black dots. (Image provided by the U.S. Centers for Disease Control and Prevention)
Though scientists and researchers are working to developing a vaccine to prevent the novel coronavirus, and drugs to treat it, those products do not currently exist. Yet some companies are selling products that purport to do just that.
Federal officials are now trying to crack down on those fraudulent products, calling their sale “a threat to the public health.”
In a statement, Dr. Stephen M. Hahn, commissioner of the U.S. Food and Drug Administration, said: “We have an aggressive surveillance program that routinely monitors online sources for health fraud products, especially during a significant public health issue such as this one.”
On Monday, the FDA and Federal Trade Commission issued warning letters to seven companies for selling fraudulent products to treat or prevent COVID-19. The agencies say those products are unapproved drugs that pose significant risks to patient health and violate federal law.
“There already is a high level of anxiety over the potential spread of coronavirus,” said FTC Chairman Joe Simons in a statement. “What we don’t need in this situation are companies preying on consumers by promoting products with fraudulent prevention and treatment claims.”
Officials are concerned that products that claim to cure, treat or prevent the virus may cause consumers to delay or stop appropriate medical treatment, leading to serious and life-threatening harm.
“We understand consumers are concerned about the spread of COVID-19 and urge them to talk to their health care providers, as well as follow advice from other federal agencies about how to prevent the spread of this illness,” Hahn said.
Warning letters were issued to the following companies: Vital Silver; Quinessence Aromatherapy Ltd.; Xephyr, LLC doing business as N-Ergetics; GuruNanda, LLC; Vivify Holistic Clinic; Herbal Amy LLC; and The Jim Bakker Show.
Among the products cited in the warning letters are teas, essential oils, tinctures and colloidal silver, which the FDA has previously said is not safe or effective for treating any disease or condition. Companies have 48 hours to respond to the letter and describe the steps they’re taking to correct the violations.
“These warning letters are just the first step. We’re prepared to take enforcement actions against companies that continue to market this type of scam,” said Simons. Companies may be subject to legal action, including but not limited to seizure or injunction.
The FDA and FTC will monitor social media, online marketplaces and incoming complaints to ensure the products aren’t sold under different company names or websites.
A task force dedicated to monitoring for fraudulent COVID-19 products has reached out to major retailers for their help in monitoring their online marketplaces for such products and has removed more than three dozen listings of fraudulent products.
Health care professionals and consumers should report adverse events or quality problems with the use of COVID-19 products to the FDA’s MedWatch Adverse Event Reporting program.
Contact Kristen Thometz: @kristenthometz | (773) 509-5452 | [email protected]